Methotrexate Toxicity
Summary
- Methotrexate-induced neurotoxicity presents with acute to subacute neurological symptoms including confusion, seizures, and focal deficits
- Pathologically characterised by demyelination and white matter injury, particularly affecting periventricular regions
- MRI demonstrates characteristic symmetric white matter T2/FLAIR hyperintensities with restricted diffusion in acute phase
Pathophysiology
- Direct toxic effect on oligodendrocytes leading to demyelination
- Disruption of folate metabolism affecting myelin synthesis and maintenance
- Accumulation of adenosine and homocysteine causing excitotoxicity
- Blood-brain barrier disruption with intrathecal administration
- Two main patterns:
- Acute/subacute leukoencephalopathy (most common)
- Chronic mineralizing microangiopathy (rare, in children)
Demographics
- Higher incidence with intrathecal administration versus systemic therapy
- Paediatric patients with ALL most commonly affected (5-10% incidence)
- Adult patients with CNS lymphoma or leptomeningeal disease
- Risk factors:
- High cumulative doses
- Concurrent cranial radiation
- Young age (<10 years) or elderly (>60 years)
- Renal insufficiency (impaired clearance)
Diagnosis
- Clinical presentation:
- Acute: confusion, somnolence, seizures (within days)
- Subacute: progressive cognitive decline, ataxia (weeks to months)
- Stroke-like episodes with focal deficits
- Transverse myelopathy (with intrathecal administration)
- Laboratory findings:
- CSF: elevated protein, mild pleocytosis
- Serum methotrexate levels (if recent administration)
- Temporal relationship to methotrexate administration crucial for diagnosis
Imaging
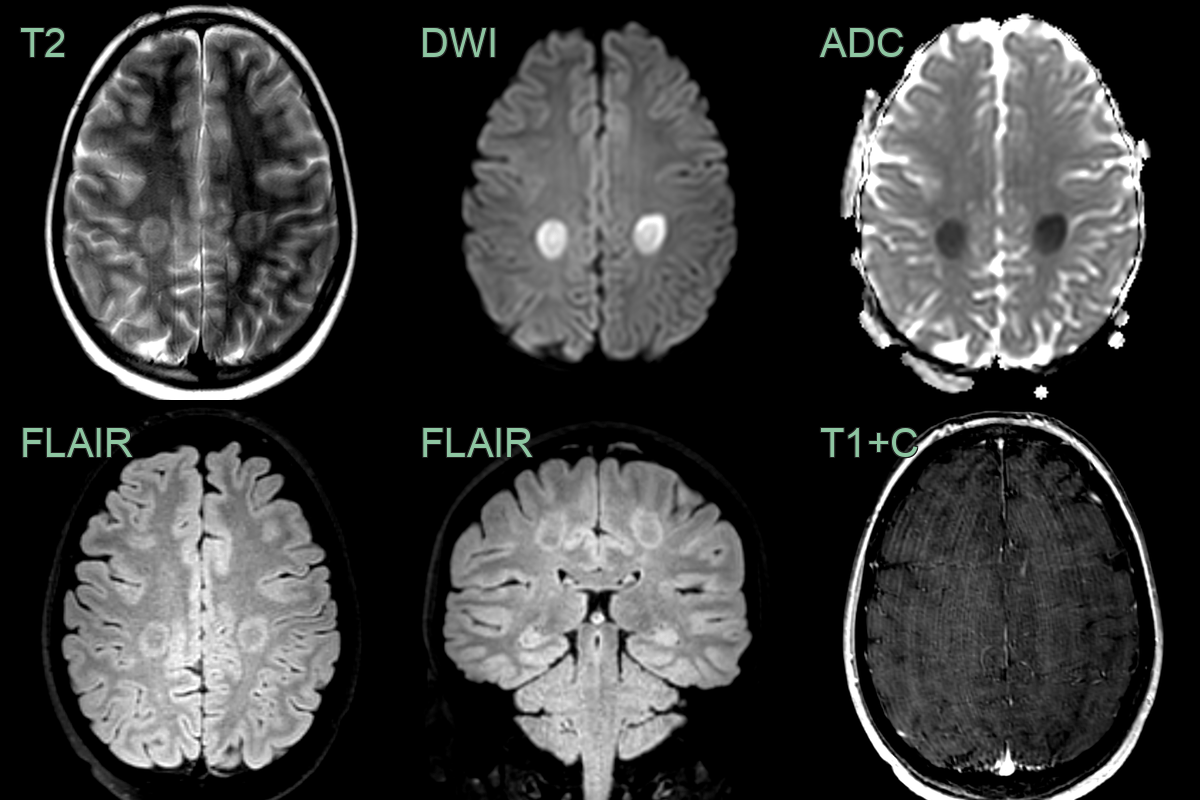
- T2/FLAIR:
- Symmetric hyperintense periventricular white matter lesions
- Centrum semiovale involvement common
- Corpus callosum may be affected
- Subcortical U-fibres typically spared
- T1:
- Hypointense in affected white matter regions
- No significant mass effect
- T1+C:
- Usually no enhancement (distinguishes from infection/tumour)
- Rare patchy enhancement in acute phase
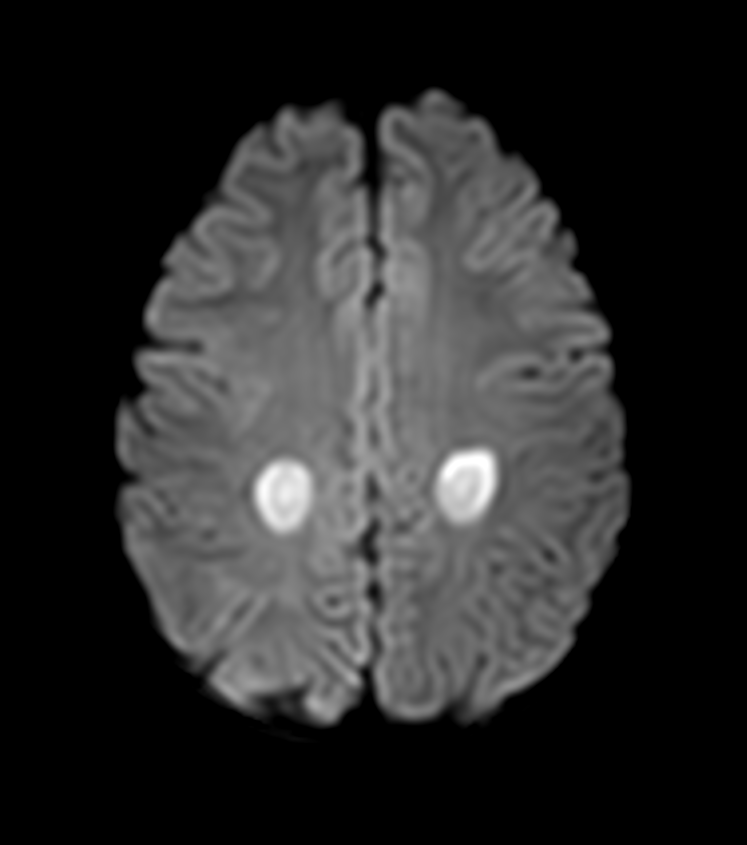
- DWI/ADC:
- Restricted diffusion (high DWI, low ADC) in acute toxicity
- Normalization or facilitated diffusion in chronic phase
- May show "reversal sign" with resolution
- SWI:
- Mineralizing microangiopathy: punctate calcifications in basal ganglia and dentate nuclei
- No haemorrhage typically
- MR Spectroscopy:
- Decreased NAA (neuronal loss)
- Elevated choline (demyelination)
- Possible lactate peak in acute phase
- CT:
- Hypodense white matter changes
- Calcifications in chronic mineralizing microangiopathy
Treatment
- Immediate discontinuation of methotrexate
- Supportive care:
- Seizure management with anticonvulsants
- Corticosteroids (controversial benefit)
- Leucovorin (folinic acid) rescue:
- 10-100 mg IV/IM every 6 hours
- Continue until methotrexate levels <0.01 μmol/L
- Aminophylline for acute toxicity (adenosine antagonist)
- Methylene blue for severe cases (experimental)
- Dextromethorphan (NMDA receptor antagonist) under investigation
- Prognosis:
- Acute toxicity often reversible with prompt treatment
- Chronic changes may be permanent
- Imaging abnormalities may persist despite clinical improvement
Differential diagnosis
| Differential diagnosis | Differentiating feature |
|---|---|
| Progressive multifocal leukoencephalopathy (PML) | Asymmetric scalloped white matter lesions with subcortical U-fibre involvement; restricted DWI at active edge; no contrast enhancement |
| ADEM | Multifocal bilateral white matter and basal ganglia lesions; ring enhancement; juxtacortical involvement |
| Multiple sclerosis | Ovoid periventricular lesions (Dawson's fingers); corpus callosum involvement; calloso-septal interface |
| Radiation-induced leukoencephalopathy | White matter changes confined to the prior radiation field |
| PRES | Posterior predominant vasogenic oedema; elevated ADC values; no corpus callosum involvement |
| HIV encephalopathy | Bilateral symmetric periventricular and subcortical white matter hyperintensities without dominant lesions |
| Osmotic demyelination syndrome | Central pontine T2 signal with trident or bat wing morphology; extrapontine basal ganglia involvement |
| Other chemotherapy-induced leukoencephalopathy | Similar periventricular white matter changes; indistinguishable from methotrexate on imaging alone |